
Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1


Chemistry, 22.06.2019 13:50
Read the chemical equation. 2c2h2 + 5o2 → 4co2 + 2h2o which of the following statements would be correct if one mole of c2h2 was used in this reaction? one mole of oxygen was used in this reaction. five moles of oxygen were used in this reaction. four moles of carbon dioxide were produced from this reaction. two moles of carbon dioxide were produced from this reaction.
Answers: 3
You know the right answer?
How many minutes are there in a solar year (365.24 days)?...
Questions

English, 22.02.2021 04:00

Mathematics, 22.02.2021 04:00


Mathematics, 22.02.2021 04:00

Advanced Placement (AP), 22.02.2021 04:00









Spanish, 22.02.2021 04:00


Mathematics, 22.02.2021 04:00


Chemistry, 22.02.2021 04:00


Chemistry, 22.02.2021 04:00

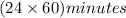 = 1440 minutes
= 1440 minutes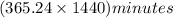 = 525945.6 minutes
= 525945.6 minutes

