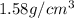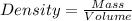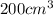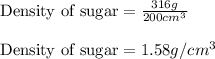Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Consider the following reaction at equilibrium. 2co2 (g) 2co (g) + o2 (g) h° = -514 kj le châtelier's principle predicts that the equilibrium partial pressure of co (g) can be maximized by carrying out the reaction a. at high temperature and high pressure b. at high temperature and low pressure c. at low temperature and low pressure d. at low temperature and high pressure e. in the presence of solid carbon
Answers: 2


Chemistry, 22.06.2019 19:50
Identify the lewis base in this balanced equation: fe3+ h2o fe(h2o)63+
Answers: 1

Chemistry, 22.06.2019 23:00
What is the mass of naoh that would have to be added to 500 ml of a solution of 0.20 m acetic acid in order to achieve a ph of 5.0?
Answers: 1
You know the right answer?
Sugar is used to fill a box that has a volume of 200 cm3. the sugar weighs 316 grams. what is the de...
Questions


English, 05.04.2021 22:10

English, 05.04.2021 22:10

Mathematics, 05.04.2021 22:10

Mathematics, 05.04.2021 22:10


Social Studies, 05.04.2021 22:10

Mathematics, 05.04.2021 22:10


English, 05.04.2021 22:10



History, 05.04.2021 22:10


Advanced Placement (AP), 05.04.2021 22:10

English, 05.04.2021 22:10



English, 05.04.2021 22:10

Geography, 05.04.2021 22:10