
Chemistry, 07.08.2019 03:10 Jazminnexoxo1093
The following thermodynamic data are available for octane, oxygen gas, carbon dioxide gas, water, and water vapor: molecule δh∘f (kj/mol) c8h18(l) −250.1 o2(g) 0 co2(g) −393.5 h2o(l) −285.8 h2o(g) −241.8 part b calculate δhrxn for the combustion of octane by using enthalpies of formation from the transition above. express the energy in kilojoules per mole to three significant figures. δhrxn δ h r x n = nothing kj/mol

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:30
How many moles of carbon dioxide will form if 2.5 moles of c3h8 is burned
Answers: 1

Chemistry, 22.06.2019 16:30
Explain in detail of the four major scientific developments that spurred the formulation of the plate tectonics theory
Answers: 2

Chemistry, 23.06.2019 00:30
What is calcium oxide+diphosphorus pentoxide--> calcium phosphate balanced
Answers: 1

You know the right answer?
The following thermodynamic data are available for octane, oxygen gas, carbon dioxide gas, water, an...
Questions

Mathematics, 06.10.2019 05:00


Chemistry, 06.10.2019 05:00


English, 06.10.2019 05:00








History, 06.10.2019 05:00

Biology, 06.10.2019 05:00

Chemistry, 06.10.2019 05:00

Mathematics, 06.10.2019 05:00


Computers and Technology, 06.10.2019 05:00


English, 06.10.2019 05:00

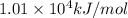
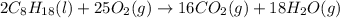
![\Delta H^o_{rxn}=\sum [n\times \Delta H^o_f(product)]-\sum [n\times \Delta H^o_f(reactant)]](/tpl/images/0172/5689/45485.png)
![\Delta H^o_{rxn}=[(16\times \Delta H^o_f_{(CO_2(g))})+(18\times \Delta H^o_f_{(H_2O(g))})]-[(2\times \Delta H^o_f_{(C_8H_{18}(l))})+(25\times \Delta H^o_f_{(O_2(g))})]](/tpl/images/0172/5689/36b5a.png)
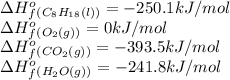
![\Delta H^o_{rxn}=[(16\times (-393.5))+(18\times (-241.8))]-[(2\times (-250.1))+(25\times (0))]=10148.2kJ/mol=1.01\times 10^4kJ/mol](/tpl/images/0172/5689/a24a9.png)


