Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:30
The following reaction shows the products when sulfuric acid and aluminum hydroxide react. al(oh)3 + h2so4 → al2(so4)3 + h2o the table shows the calculated amounts of reactants and products when the reaction was conducted in a laboratory. sulfuric acid aluminum hydroxide initial amount of reactant 40 g 15 g theoretical yield of water from reactant 14.69 g 10.38 g what is the approximate amount of the leftover reactant? 11.73 g of sulfuric acid 10.33 g of sulfuric acid 11.12 g of aluminum hydroxide 13.67 g of aluminum hydroxide
Answers: 3

Chemistry, 22.06.2019 08:30
What is the independent variable in this investigation? mass volume sample number substance density
Answers: 3

Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3
You know the right answer?
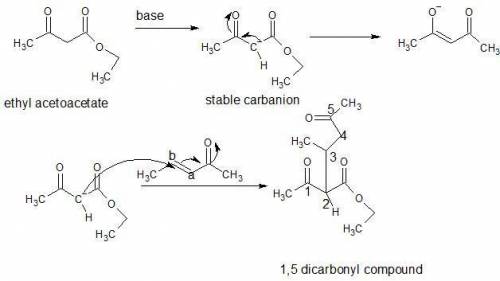
In a michael reaction, an a carbon attacks the b carbon of an a, b-unsaturated carbonyl compound to...
Questions

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01

World Languages, 11.09.2020 16:01

Social Studies, 11.09.2020 16:01

Biology, 11.09.2020 16:01

Mathematics, 11.09.2020 16:01