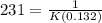Aparticular reactant decomposes with a half‑life of 109 s when its initial concentration is 0.280 m. the same reactant decomposes with a half‑life of 231 s when its initial concentration is 0.132 m.
1. determine the reaction order.
(a)1
(b)2
(c)0
2. what is the value and units of the rate constant for this reaction? =

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Drive down any three characteristic of modern periodic table
Answers: 1

Chemistry, 22.06.2019 02:30
Margaret wants to make an orange flavored drink by stirring powdered drink mix into a glass of water. she doesn't like drinks that have small clumps of powdered solid in them, so she wants the drink to be a perfect solution. what factors should margaret not consider when deciding how much powder to add to her glass of water?
Answers: 3

Chemistry, 22.06.2019 05:30
A3.37-mg sample of protein was chemically digested to convert its nitrogen into ammonia and then diluted to 100.0 ml. then 10.0 ml of this solution was placed in a 50-ml volumetric flask and treated with 5 ml of phenol solution plus 2 ml of sodium hypochlorite solution. the sample was diluted to 50.0 ml, and the absorbance at 625 nm was measured in a 1.00-cm cuvette and found to be 0.486. for reference, a standard solution was prepared from 10.0 mg of nh4cl (molar mass = 53.49 grams/mole) dissolved in 1.00 l of water. then 10.0 ml of this standard was placed in a 50-ml volumetric flask, treated in the same manner as the unknown, and the absorbance found to be 0.323. finally, a reagent blank was prepared using distilled water in place of unknown, it was treated in the same manner as the unknown, and the absorbance found to be 0.076. calculate the weight percent of nitrogen in the protein.
Answers: 1

Chemistry, 22.06.2019 08:30
In a chemical reaction at equilibrium, the rate of the forward reaction the rate of the reverse reaction. if the rate of the forward reaction more products are formed.
Answers: 1
You know the right answer?
Aparticular reactant decomposes with a half‑life of 109 s when its initial concentration is 0.280 m....
Questions

History, 22.04.2021 05:00



Mathematics, 22.04.2021 05:00


Mathematics, 22.04.2021 05:00



Computers and Technology, 22.04.2021 05:00






Spanish, 22.04.2021 05:00

English, 22.04.2021 05:00


Physics, 22.04.2021 05:00


![\frac{1}{k[A_{0}]^{(n-1)}}](/tpl/images/0155/1180/be91f.png)
![K\frac{1}{[A_{0}]^{(n-1)} }](/tpl/images/0155/1180/1baec.png)
![\frac{(halflife_{1})}{(halflife_{2})}=\frac{[A_{2}]^{(n-1)}}{[A_{1}]^{(n-1)} }](/tpl/images/0155/1180/93500.png)
![\frac{109}{231}=\frac{[0.132]^{(n-1)}}{[0.280]^{(n-1)}}](/tpl/images/0155/1180/d6c94.png)
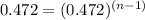
![halflife=\frac{1}{k[A_{0}]}](/tpl/images/0155/1180/b0fd9.png)