Chemistry, 19.07.2019 01:30 JakerGamer
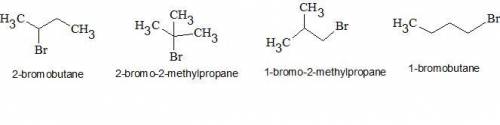
7.50 draw all constitutional isomers with the molecular formula c4h9br, and then arrange them in order of: (a) increasing reactivity toward an sn2 reaction. (b) increasing reactivity toward an e2 reaction

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Abullet found at a crime scene may be used as evidence in a trial if the percentage of metals match to the composition of metals in a bullet from the suspect's ammunition. a forensic scientist's analysis of the bullet shows that it contains 11.9 g of lead, 0.5 g of tin, and 0.8 b of antimony. what is the percentage of lead metal in the bullet? express your answers to the one's place.
Answers: 2

Chemistry, 21.06.2019 20:00
Different isotopes indicate that an element will have different numbers of
Answers: 2

Chemistry, 21.06.2019 23:20
Harvey kept a balloon with a volume of 348 milliliters at 25.0˚c inside a freezer for a night. when he took it out, its new volume was 322 milliliters, but its pressure was the same. if the final temperature of the balloon is the same as the freezer’s, what is the temperature of the freezer? the temperature of the freezer is kelvins.
Answers: 2

Chemistry, 22.06.2019 10:50
A100 kmol/h stream that is 97 mole% carbon tetrachloride (ccl4) and 3% carbon disulfide (cs2) is to be recovered from the bottom of a distillation column. the feed to the column is 16 mole% cs2 and 84% ccl4, and 2% of the ccl4 entering the column is contained in the overhead stream leaving the top of the column. calculate the mass and mole fractions of ccl4 in the overhead stream, and determine the molar flow rates of ccl4 and cs2 in the overhead and feed streams. 12. mw_ccla- 153.82; mw_cs2-76.14.
Answers: 3
You know the right answer?
7.50 draw all constitutional isomers with the molecular formula c4h9br, and then arrange them in ord...
Questions

Computers and Technology, 02.12.2021 04:30




Biology, 02.12.2021 04:30





English, 02.12.2021 04:30

Computers and Technology, 02.12.2021 04:30

History, 02.12.2021 04:30

Mathematics, 02.12.2021 04:30





Social Studies, 02.12.2021 04:30

Mathematics, 02.12.2021 04:30