Which statement best describes balancing equations and the law of conservation of mass?
there...

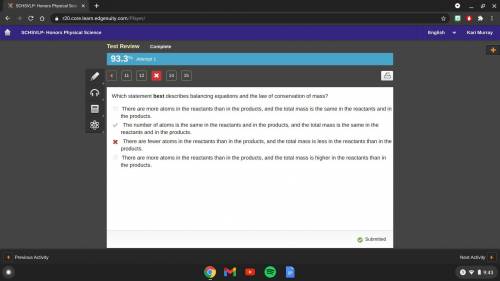
Which statement best describes balancing equations and the law of conservation of mass?
there are more atoms in the reactants than in the products, and the total mass is the same in the reactants and in the
products
the number of atoms is the same in the reactants and in the products, and the total mass is the same in the reactants and
in the products
there are fewer atoms in the reactants than in the products, and the total mass is less in the reactants than in the
products
there are more atoms in the reactants than in the products, and the total mass is higher in the reactants than in the
products

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:00
How is energy related to the change of state represented by the model? atoms gain energy as a solid changes to a liquid. atoms gain energy as a solid changes to a gas. atoms lose energy as a solid changes to a liquid. atoms lose energy as a solid changes to a gas.
Answers: 3

Chemistry, 22.06.2019 19:20
Consider hydrogen in an excited state n = 5n=5 that emits photons to reach the ground state. there are various possible transitions other than straight to the ground state that can occur; for example, it can drop to the n = 3n=3 state followed by the n = 3n=3 to the ground state transition. which of the possible transitions will result in the emission of a photon in the visible region?
Answers: 3

Chemistry, 22.06.2019 21:00
Write a balanced equation showing the formation of copper (ii) nitrite from its elements
Answers: 1

Chemistry, 23.06.2019 02:40
Calculate the standard enthalpy of formation of liquid methanol, ch3oh(l), using the following information: c(graphite) + o2 latex: \longrightarrow ⟶ co2(g) latex: \delta δ h° = –393.5 kj/mol h2(g) + o2 latex: \longrightarrow ⟶ h2o(l) latex: \delta δ h° = –285.8 kj/mol ch3oh(l) + o2(g) latex: \longrightarrow ⟶ co2(g) + 2h2o(l) latex: \delta δ h° = –726.4 kj/mol
Answers: 3
You know the right answer?
Questions

Health, 15.10.2019 22:30

Biology, 15.10.2019 22:30

Geography, 15.10.2019 22:30

Social Studies, 15.10.2019 22:30

Arts, 15.10.2019 22:30

History, 15.10.2019 22:30


Computers and Technology, 15.10.2019 22:30


Mathematics, 15.10.2019 22:30

English, 15.10.2019 22:30

Health, 15.10.2019 22:30

History, 15.10.2019 22:30

Mathematics, 15.10.2019 22:30


Biology, 15.10.2019 22:30



Mathematics, 15.10.2019 22:30