
Chemistry, 10.10.2019 12:00 savannahvargas512
Compounds a and b react to form compounds c and d according to the equation: aa + bb → cc + dd. under which conditions will the rate law be given by the equation: rate = k[a]a[b]b? a. the reaction takes place in one step. b. the reaction is endothermic. c. the reaction is exothermic. d. the reaction involves more than one step.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 05:40
Consider the elements bromine and chlorine; which elements has a larger ionic radius ?
Answers: 1

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 11:00
Which statement is true about hcl? (5 points) select one: a. it is a salt because it increases the concentration of metallic ions. b. it is a salt because it is formed by the reaction of an acid and a base. c. it is an acid because it increases the concentration of hydroxyl ions. d. it is an acid because it increases the concentration of hydronium ions.
Answers: 1
You know the right answer?
Compounds a and b react to form compounds c and d according to the equation: aa + bb → cc + dd. und...
Questions

History, 25.07.2019 17:20

Mathematics, 25.07.2019 17:20

Mathematics, 25.07.2019 17:20

Chemistry, 25.07.2019 17:20



History, 25.07.2019 17:20

Business, 25.07.2019 17:20

Social Studies, 25.07.2019 17:20

History, 25.07.2019 17:20

History, 25.07.2019 17:20

Biology, 25.07.2019 17:20

Social Studies, 25.07.2019 17:20

Biology, 25.07.2019 17:20






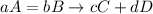
![Rate=k[A]^a[B]^b](/tpl/images/0306/8163/8c0b1.png)


