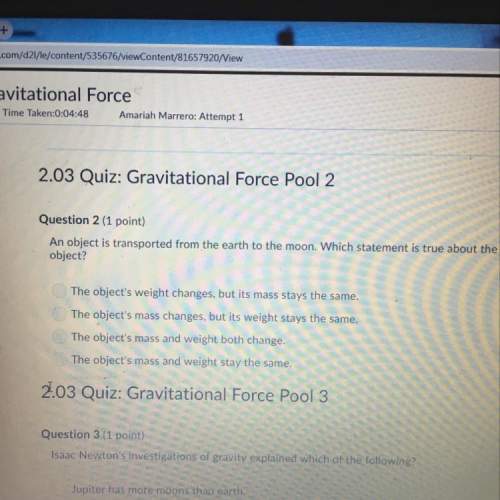
Chemistry, 06.07.2019 01:10 priceisright11401
2n h 3 ( g ) ⟷ n 2 ( g ) + 3 h 2 ( g ) k p = 0.83 consider your answers above, if the initial pressures for all three species is 1 atm what is the equilibrium pressure of h2? (hint: your quadratic will have two solutions, which one is impossible? )

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Which is true of a liquid? it has a definite volume but not a definite mass.it has a definite mass but not a definite volume.it has a definite volume but not a definite shape.it has a definite shape but not a definite volume.
Answers: 2

Chemistry, 22.06.2019 17:00
Reduction is a reaction which results in a in electrons and a in positive charge of the atom or ion 1) a- loss 1) b- gain 2) a-increase 2) b-decrease
Answers: 1

Chemistry, 22.06.2019 17:30
To find the enthalpy of a reaction in the lab, you measured the of the reactants and the change during the reaction.
Answers: 1

Chemistry, 22.06.2019 18:10
Consider the following reaction at equilibrium: c(s)+h2o(g)⇌co(g)+h2(g) predict whether the reaction will shift left, shift right, or remain unchanged upon each of the following disturbances. a) c is added to the reaction mixture. b) h2ois condensed and removed from the reaction mixture c) co is added to the reaction mixture d) h2 is removed from the reaction mixture.
Answers: 3
You know the right answer?
2n h 3 ( g ) ⟷ n 2 ( g ) + 3 h 2 ( g ) k p = 0.83 consider your answers above, if the initial pressu...
Questions





Computers and Technology, 03.11.2020 18:20

History, 03.11.2020 18:20


History, 03.11.2020 18:20


Arts, 03.11.2020 18:20


History, 03.11.2020 18:20


Engineering, 03.11.2020 18:20

Social Studies, 03.11.2020 18:20

English, 03.11.2020 18:20

Biology, 03.11.2020 18:20


Biology, 03.11.2020 18:20

Social Studies, 03.11.2020 18:20