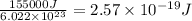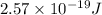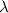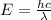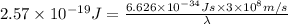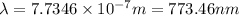Chemistry, 06.07.2019 00:30 Person51761
It takes 155. kj/mol to break a fluorine-fluorine single bond. calculate the maximum wavelength of light for which a flouine-flouring single bond could be broken by absorbing a single photon

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:00
Will give brainliest it is a lab from k12 here is the linkfor each metal that participated in a chemical change, write the type of metal it is, based on your examination of the periodic table. type your answer here. (score for question 3: of 5 points) were there any metallic compounds that did not react with either the acid or the base? write the type of metal, based on your examination of the periodic table. type your answer here. (score for question 4: of 5 points) make a general statement about the reactivity of the metals in this experiment. type your answer here.
Answers: 2

Chemistry, 22.06.2019 03:30
In saturated organic compounds, all the bonds between carbon atoms are called?
Answers: 1

Chemistry, 22.06.2019 05:30
Compare and contrast physical changes with chemical changes.
Answers: 1

Chemistry, 22.06.2019 07:00
6what is the importance of water on earth? a) it keeps the top layer of the geosphere cool b) it allows life to exist c) it provides ice at the poles d) it creates earth's blue color from space
Answers: 2
You know the right answer?
It takes 155. kj/mol to break a fluorine-fluorine single bond. calculate the maximum wavelength of l...
Questions

History, 18.10.2020 06:01

Social Studies, 18.10.2020 06:01

Mathematics, 18.10.2020 06:01

Spanish, 18.10.2020 06:01

Mathematics, 18.10.2020 06:01

Mathematics, 18.10.2020 06:01



History, 18.10.2020 06:01



Mathematics, 18.10.2020 06:01


English, 18.10.2020 06:01

Mathematics, 18.10.2020 06:01



Social Studies, 18.10.2020 06:01

Mathematics, 18.10.2020 06:01

Social Studies, 18.10.2020 06:01

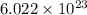 molecules = 155000 J
molecules = 155000 J