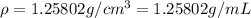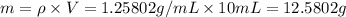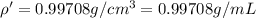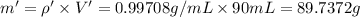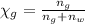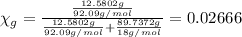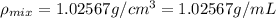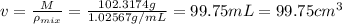You mix 10 ml glycerol and 90 ml water to obtain a 10% glycerol solution. the density of the mixture is ρmix = 1.02567 g/cm. what are the mole fraction of glycerol and the volume of the mixture? what is the reason for the volume change? mm(glycerol) = 92.09 g/mol, mm(h2o) = 18 g/mol, ρ(glycerol) = 1.25802 g/cm3, ρ(h2o) = 0.99708 g/cm3.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
Explain why scientists use shared characteristics to make cladograms.
Answers: 1

Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 10:10
Stage in which a star’s outer layers have started to cool and grow outward?
Answers: 3
You know the right answer?
You mix 10 ml glycerol and 90 ml water to obtain a 10% glycerol solution. the density of the mixture...
Questions

Mathematics, 24.11.2019 00:31


English, 24.11.2019 00:31


History, 24.11.2019 00:31



Mathematics, 24.11.2019 00:31



Health, 24.11.2019 00:31

Biology, 24.11.2019 00:31

Mathematics, 24.11.2019 00:31

Biology, 24.11.2019 00:31



Mathematics, 24.11.2019 01:31

Social Studies, 24.11.2019 01:31