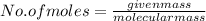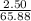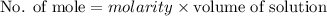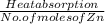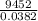Chemistry, 29.06.2019 00:20 webbjalia04
A2.50 g sample of powdered zinc is added to 100.0 ml of a 2.00 m aqueous solution of hydrobromic acid in a calorimeter. the total heat capacity of the calorimeter and solution is 448 j/k. the observed increase in temperature is 21.1 k at a constant pressure of one bar. calculate the standard enthalpy of reaction using these data. zn(s)+2hbr(aq)⟶znbr2(aq)+h2(g)

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 23:30
Write a paragraph that provides examples of each stage of volcanic activity, a description of the volcano, and facts about each stage.
Answers: 1

Chemistry, 22.06.2019 13:30
Which statements are true concerning mineral formation? check all that apply. the slower the cooling, the larger the crystals. the faster the cooling, the smaller the crystals. crystals formed from magma are smaller than crystals formed from lava. minerals can only form in solutions when the solution is heated deep underground. when a solution cools, elements and compounds leave the solution and crystallize as minerals. minerals formed from hot water solutions can form narrow channels in the surrounding rock.
Answers: 1

Chemistry, 22.06.2019 22:30
The vapor pressure of ethanol is 1.00 × 102 mmhg at 34.90°c. what is its vapor pressure at 61.61°c? (δhvap for ethanol is 39.3 kj/mol.)
Answers: 2

Chemistry, 22.06.2019 23:00
What is the formula of the ionic compound composed of calcium cations and chloride anions
Answers: 1
You know the right answer?
A2.50 g sample of powdered zinc is added to 100.0 ml of a 2.00 m aqueous solution of hydrobromic aci...
Questions



Social Studies, 31.01.2020 04:45

English, 31.01.2020 04:45

Social Studies, 31.01.2020 04:45

Mathematics, 31.01.2020 04:45

Mathematics, 31.01.2020 04:45


History, 31.01.2020 04:45


Biology, 31.01.2020 04:45



Mathematics, 31.01.2020 04:45


Mathematics, 31.01.2020 04:45