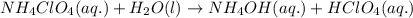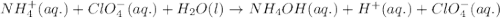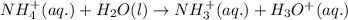Chemistry, 05.12.2019 18:31 naynay1997
Write the net ionic equation for the acid-base hydrolysis equilibrium that is established when ammonium perchlorate is dissolved in water. (use h3o+ instead of h+.)

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 12:00
An atom's configuration based on its number of electrons ends at 3p4. another atom has seven more electrons. starting at 3p, what is the remaining configuration? 3p63d34s2 3p43d54s2 3p64s23d3 3p44s23d
Answers: 3


Chemistry, 23.06.2019 07:30
The compound formed from 2 atoms of hydrogen and one atom of oxygen is
Answers: 1
You know the right answer?
Write the net ionic equation for the acid-base hydrolysis equilibrium that is established when ammon...
Questions




Biology, 19.02.2021 05:00

Mathematics, 19.02.2021 05:00

Mathematics, 19.02.2021 05:00


Mathematics, 19.02.2021 05:00

Physics, 19.02.2021 05:00

Chemistry, 19.02.2021 05:00


Mathematics, 19.02.2021 05:00







Biology, 19.02.2021 05:00