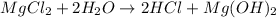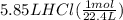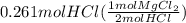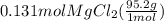Chemistry, 21.12.2019 04:31 kailahgranger
When magnesium chloride reacts with water, 5.85 l hcl(g) is produced.
molar mass of hcl = 36.5 g/mol
molar mass of mgcl2 = 95.2 g/mol
how many moles of hcl was produced?
how many moles of mgcl2 reacted?
what mass of mgcl2 reacted?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 09:00
The nuclear fission process releases neutrons and question 27 options: alpha particles electrons energy beta particles
Answers: 1

Chemistry, 23.06.2019 03:30
Select the correct lewis structure for fluorine which is group 7a element?
Answers: 1

Chemistry, 23.06.2019 04:31
Use the drop-down menus to label each of the following changes p for physical change and c for chemical change. the substance changes to a new substance. the original substance can be recovered. the color changes. gas is produced and given off. the substance changes size, shape, or volume.
Answers: 2

Chemistry, 23.06.2019 13:30
1. what is boyle’s law? • state the definition of the law in words. • what are the assumptions of boyle’s law? • write at least one mathematical equation that represents the law. • what can be calculated with boyle’s law? • using a gas-filled balloon as an example, describe what is happening to the gas molecules inside the balloon before and after you squeeze it.
Answers: 2
You know the right answer?
When magnesium chloride reacts with water, 5.85 l hcl(g) is produced.
molar mass of hcl = 36....
molar mass of hcl = 36....
Questions


History, 19.08.2019 22:50

Computers and Technology, 19.08.2019 22:50

English, 19.08.2019 22:50


History, 19.08.2019 22:50

Social Studies, 19.08.2019 22:50



Mathematics, 19.08.2019 22:50

Physics, 19.08.2019 22:50

Mathematics, 19.08.2019 22:50

Mathematics, 19.08.2019 22:50


Spanish, 19.08.2019 22:50




Social Studies, 19.08.2019 22:50

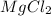 and (iii) 12.5 grams of
and (iii) 12.5 grams of