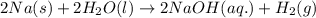Chemistry, 21.06.2019 15:50 algahimnada
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hydrogen gas and aqueous sodium hydroxide. part a write a balanced chemical equation for this reaction. express your answer as a chemical equation. identify all of the phases in your answer.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 12:30
Consider the four elements above. which one of these elements will combine with oxygen in a 1: 1 ratio?
Answers: 3

Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2
You know the right answer?
In a popular classroom demonstration, solid sodium is added to liquid water and reacts to produce hy...
Questions


Mathematics, 31.08.2021 06:40


Mathematics, 31.08.2021 06:40

Health, 31.08.2021 06:40

Mathematics, 31.08.2021 06:40



Mathematics, 31.08.2021 06:40

English, 31.08.2021 06:40

Geography, 31.08.2021 06:40






English, 31.08.2021 06:40


Mathematics, 31.08.2021 06:40

English, 31.08.2021 06:40