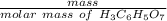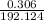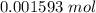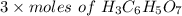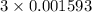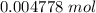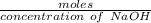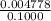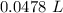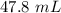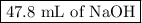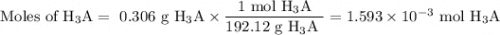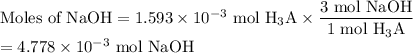Chemistry, 22.06.2019 12:20 missayers172
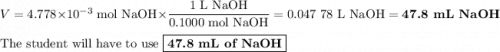
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml volumetric flask and dilutes to the mark with distilled water. he plans to titrate the acid with 0.1000 m naoh solution. calculate the volume of naoh solution the student will need to add to reach the final equivalence point. be sure your answer has the correct number of significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:30
Calculate the change in entropy if br2(l) is converted to br2(g). s° for br2(l) = 152.2 j/(mol•k) s° for br2(g) = 245.5 j/(mol•k) s° for br(g) = 175.0 j/(mol•k)
Answers: 3

Chemistry, 21.06.2019 21:30
Aphysical reaction is a process in which one or more reactants change into one or more products with different properties. select the best answer from the choices provided t f
Answers: 1


Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3
You know the right answer?
Achemistry student weighs out 0.306 g of citric acid (h3c6h5o7), a triprotic acid, into a 250 ml vol...
Questions

Mathematics, 29.06.2019 05:00

English, 29.06.2019 05:00

Mathematics, 29.06.2019 05:00


Mathematics, 29.06.2019 05:00

Mathematics, 29.06.2019 05:00




Geography, 29.06.2019 05:00

Social Studies, 29.06.2019 05:00


History, 29.06.2019 05:00




History, 29.06.2019 05:00

History, 29.06.2019 05:00

History, 29.06.2019 05:00

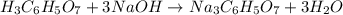
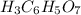 will be:
will be: