
Chemistry, 27.06.2019 14:50 MrKrinkle77
Question 7(multiple choice worth 4 points) (08.05 mc) what is the ph of a solution with a 3.8 × 10−4 m hydronium ion concentration? 3.4 3.8 11 12

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:00
You encounter a solution that is acidic and you decide to test it by adding a small amount of a strong acid. the ph lowers slightly but is approximately unchanged, and still remains acidic. what can you say about the solution? a. it is a buffer solution. b. it is not a buffer solution it is a strong acid solution. d. the solution has been neutralized. e. the solution has excess acid present
Answers: 1

Chemistry, 22.06.2019 13:00
The molality of calcium chloride (cacl2) in an aqueous solution is 2.46 m. what is mole fraction of the solute?
Answers: 3

Chemistry, 22.06.2019 18:30
What volume of a 0.0606 m solution of strontium bromide is needed to obtain 0.340 mol of the compound? question 42 options: a)5.61 l b) 3.4 l c) 600 ml d) 1 l e) 178 ml
Answers: 1

Chemistry, 22.06.2019 23:00
How does the value of the equilibrium constant show that a reaction reaches equilibrium very quickly? (a) the equilibrium constant is large. (b) the equilibrium constant is small. (c) the equilibrium constant is zero. (d) the value of the equilibrium constant does not show how quickly a reaction comes to equilibrium.
Answers: 1
You know the right answer?
Question 7(multiple choice worth 4 points) (08.05 mc) what is the ph of a solution with a 3.8 × 10−...
Questions

Mathematics, 01.02.2022 18:30

Mathematics, 01.02.2022 18:30

Spanish, 01.02.2022 18:30

Mathematics, 01.02.2022 18:30

Chemistry, 01.02.2022 18:30


Mathematics, 01.02.2022 18:30


Biology, 01.02.2022 18:30


Social Studies, 01.02.2022 18:30



Mathematics, 01.02.2022 18:30


Geography, 01.02.2022 18:30



Physics, 01.02.2022 18:30

![\displaystyle \rm pH = \log{\frac{1}{[H_{3}O^{+}]}} = - \log{[H_3O}^{+}]}](/tpl/images/0023/6873/cb610.png) ,
,![\rm [H_3O}^{+}]](/tpl/images/0023/6873/23038.png) is the hydronium ion concentration of the solution in moles per liter. Note that some textbooks write
is the hydronium ion concentration of the solution in moles per liter. Note that some textbooks write 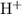 .
. 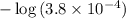
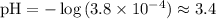 .
.

