How many grams of nahco3 would you need to react with 6 moles of h2so4?
the balanced chemica...

Chemistry, 26.10.2019 23:43 ewalchloe5067920
How many grams of nahco3 would you need to react with 6 moles of h2so4?
the balanced chemical equation is:
h2so4(aq) + 2 nahco3 (s) —-> na2so4 (aq) + 2 co2 (g) + 2 h2o

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Liv sheldon given the balanced equation for an organic reaction: c2h2 + 2cl2 → c2h2cl4 this reaction is best classified as *
Answers: 1

Chemistry, 22.06.2019 06:00
How much would the freezing point of water decrease if 4 mol of sugar were added to 1 kg of water(k=1.86 c/mol/kg for water and i=1 for sugar
Answers: 1

Chemistry, 22.06.2019 12:30
What metric units would you use to measure the thickness of a key
Answers: 3

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1
You know the right answer?
Questions



Mathematics, 23.02.2022 20:20

Mathematics, 23.02.2022 20:20


English, 23.02.2022 20:20


Business, 23.02.2022 20:20






Engineering, 23.02.2022 20:20

Computers and Technology, 23.02.2022 20:20


Mathematics, 23.02.2022 20:20


Mathematics, 23.02.2022 20:20

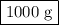
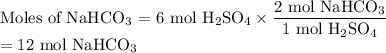
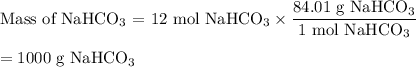
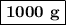 of NaHCO₃.
of NaHCO₃.

