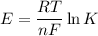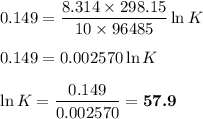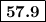Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:00
0.66y = 0.9x + 0.48 if y has a value of 108.45 what is the value of x?
Answers: 1

Chemistry, 22.06.2019 00:30
Asolution of sodium hydroxide was titrated against a solution of sulfuric acid. how many moles of sodium hydroxide would react with 1 mole of sulfuric acid?
Answers: 2

Chemistry, 22.06.2019 14:30
Calculate the mass of carbon in 97.0 g of sucrose c12h22o11
Answers: 3

Chemistry, 22.06.2019 17:30
Consider the story you just read. all but one of the choices below indicate that something is living.
Answers: 1
You know the right answer?
From standard reduction potentials, calculate the equilibrium constant at 25 ∘c for the reaction 2mn...
Questions

Social Studies, 04.11.2020 22:30



Mathematics, 04.11.2020 22:30







History, 04.11.2020 22:30



Mathematics, 04.11.2020 22:30

Advanced Placement (AP), 04.11.2020 22:30

Mathematics, 04.11.2020 22:30



Mathematics, 04.11.2020 22:30