
Chemistry, 08.01.2020 10:31 mandilynn22
How many moles of sulfur atoms are present in 2.00 moles of cs2 ?

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Sex cells from female reproductive organ? 1) mitosis 2) fertilization 3) zygote 4) eggs 5) meiosis 6) sperm
Answers: 2

Chemistry, 21.06.2019 21:40
During trial 2, what allowed you to determine that aluminum was the limiting reactant? check all that apply. all of the copper dissolved. all of the aluminum dissolved. the solution turned clear. the number of grams of copper(ii) chloride used in the reaction was greater than the number of grams of aluminum. the molar ratio of copper(ii) chloride to aluminum was greater than 3: 2, the equation’s molar ratio.
Answers: 2

Chemistry, 22.06.2019 01:50
Ase your answer to this question on the information below.hydrocarbons and fissionable nuclei are among the sources used for the production of energy in the united states. a chemical reaction produces much less energy than a nuclear reaction per mole of reactant.the balanced chemical equation below represents the reaction of one molecule of a hydrocarbon with two molecules of oxygen.chemical equation: ch4 + 2o2 → co2 + 2h2o + 1.48 × 10−18 jthe nuclear equation below represents one of the many possible reactions for one fissionable nucleus. in this equation, x represents a missing product.nuclear equation: write an isotopic notation for the missing product represented by x in the nuclear equation.
Answers: 1

Chemistry, 22.06.2019 08:20
What is the formula for the compound dinitrogen pentoxide? a. n4o5 b. n5o4 c. n4o6 d. n5o2 e. n2o5
Answers: 3
You know the right answer?
How many moles of sulfur atoms are present in 2.00 moles of cs2 ?...
Questions

Business, 13.07.2019 14:30


Mathematics, 13.07.2019 14:30

Mathematics, 13.07.2019 14:30


Mathematics, 13.07.2019 14:30

Biology, 13.07.2019 14:30

Mathematics, 13.07.2019 14:30


Mathematics, 13.07.2019 14:30

Mathematics, 13.07.2019 14:30

Mathematics, 13.07.2019 14:30


Mathematics, 13.07.2019 14:30

SAT, 13.07.2019 14:30


Mathematics, 13.07.2019 14:30

Mathematics, 13.07.2019 14:30

History, 13.07.2019 14:30

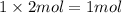 of carbon atom
of carbon atom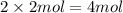 of sulfur atom
of sulfur atom

