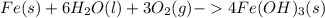One of the most recognizable corrosion reactions is the rusting of iron. rust is caused by iron reacting with oxygen gas in the presence of water to create an oxide layer. iron can form several different oxides, each having its own unique color. red rust is caused by the formation of iron(iii) oxide trihydrate. in the space provided, write the balanced reaction for the formation of fe2o3•3h2o(s). phases are optional.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 03:30
In this chemical reaction, 325 grams of barium (ba) react completely. how many moles of lithium (li) are produced?
Answers: 1

Chemistry, 22.06.2019 12:00
Hey guys so i need to know what is _nh3+> nh4oh ~chemistry~
Answers: 1

Chemistry, 23.06.2019 00:10
Find the missing probability in the table below a.0.10 b.40 c.0.80 d. 0.20
Answers: 2

Chemistry, 23.06.2019 05:30
For the reaction i2(g)+br2(g)←−→2ibr(g), kc=280 at 150 ∘c. suppose that 0.450 mol ibr in a 2.00-l flask is allowed to reach equilibrium at 150 ∘c. what is the equilibrium concentration of 2ibr, i2, br2
Answers: 1
You know the right answer?
One of the most recognizable corrosion reactions is the rusting of iron. rust is caused by iron reac...
Questions


Mathematics, 26.09.2021 19:10



Mathematics, 26.09.2021 19:10


Biology, 26.09.2021 19:10


Biology, 26.09.2021 19:10






Biology, 26.09.2021 19:10


Engineering, 26.09.2021 19:10

Mathematics, 26.09.2021 19:10