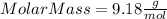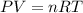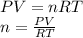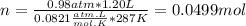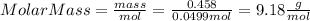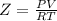Part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1.20 l at 287 k and 0.980 atm. show your work. part 2. if this sample was placed under extreme pressure, describe how the actual volume would compare to the predicted volume. explain your answer.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 11:30
Compare and contrast refraction of light and sound will give brainliest
Answers: 1

Chemistry, 22.06.2019 13:00
Using the thermodynamic information in the aleks data tab, calculate the standard reaction free energy of the following chemical reaction: →+p4o10s6h2ol4h3po4s round your answer to zero decimal places.
Answers: 3

Chemistry, 22.06.2019 14:30
Which of the following describes a situation where competition between producers exists
Answers: 1

Chemistry, 23.06.2019 05:00
Match the term to its description match term definition chemical energy a) internal energy caused by vibrations of atoms and molecules electrical energy b) electromagnetic energy that travels in waves radiant energy c) the movement of an electrical charge thermal energy d) potential energy stored in the bonds between atoms
Answers: 1
You know the right answer?
Part 1. determine the molar mass of a 0.458-gram sample of gas having a volume of 1.20 l at 287 k an...
Questions


Mathematics, 20.08.2019 02:40

Social Studies, 20.08.2019 02:40




Mathematics, 20.08.2019 02:40

Mathematics, 20.08.2019 02:40

Geography, 20.08.2019 02:40







Social Studies, 20.08.2019 02:40

Mathematics, 20.08.2019 02:40