

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
Determine the empirical formula of a compound containing 40.6 grams of carbon, 5.1 grams of hydrogen, and 54.2 grams of oxygen. in an experiment, the molar mass of the compound was determined to be 118.084 g/mol. what is the molecular formula of the compound? for both questions, show your work or explain how you determined the formulas by giving specific values used in calculations.
Answers: 3

Chemistry, 22.06.2019 03:50
What is the temperature of one mole of helium gas at stp?
Answers: 3

Chemistry, 22.06.2019 10:10
What shape would a molecule with two bound groups and two lone pairs have?
Answers: 1

Chemistry, 22.06.2019 18:30
Asample of hydrated tin (ii) chloride (sncl2) has a mass of 4.90 g. when it is dehydrated, it has a mass of 4.10 g. which is the correct chemical formula for the hydrate? sncl2•2h2o sncl2•4h2o sncl2•6h2o
Answers: 2
You know the right answer?
What is the mass present in a 10.0l container of oxygen at a pressure of 105kpa and 20 degrees celsi...
Questions


Mathematics, 14.09.2021 14:00

History, 14.09.2021 14:00



Mathematics, 14.09.2021 14:00


Mathematics, 14.09.2021 14:00



Mathematics, 14.09.2021 14:00

Mathematics, 14.09.2021 14:00


Mathematics, 14.09.2021 14:00

Mathematics, 14.09.2021 14:00



Mathematics, 14.09.2021 14:00


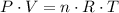 ,
, 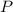 the pressure on the gas,
the pressure on the gas, 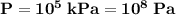 ;
;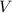 the volume of the gas,
the volume of the gas, 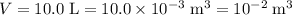 ;
;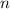 the number of moles of the gas, which needs to be found;
the number of moles of the gas, which needs to be found;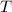 the absolute temperature of the gas,
the absolute temperature of the gas, 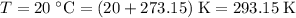 .
.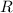 the ideal gas constant,
the ideal gas constant, 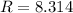 if P, V, and T are in their corresponding SI units: Pa, m³, and K.
if P, V, and T are in their corresponding SI units: Pa, m³, and K.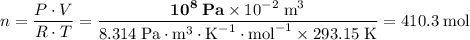 .
.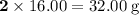 . The mass of 410.3 moles of O₂ will be:
. The mass of 410.3 moles of O₂ will be: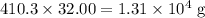 .
.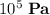 or
or 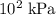 instead?
instead?

