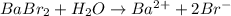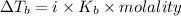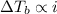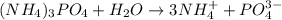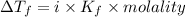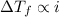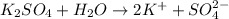Chemistry, 03.02.2020 06:02 Teenabrewer28
Ineed someone to see if my answers are correct!
1. for solutions, colligative properties depend only on the number of dissolved particles in a given volume and do not depend on the identity of the solute.
a. true
b. false < my choice
2. what number of particles will nh3 make in a solution.
a. 1
b. 2 < my choice
c. 3
d. 4
3. what number of particles will c6h12 make in solution.
a. 1
b. 2
c. 3
d. 4 < my choice
4. boiling occurs when the vapor pressure of a liquid equals the atmospheric pressure. in the high altitude city of denver, the boiling point of water is
a. > 100°c < my choice
b. 100°c
c. < 100°c
5. what number of particles will al(no3) 3 make in solution
a. 1 < my choice
b. 2
c. 3
d. 4
6. what number of particles will babr2 make in solution.
a. 1
b. 2
c. 3 < my choice
d. 4
7. determine which compound would raise the boiling point of water the most.
a. 2.0 m (nh4)3po4
b. 2.0 m nacl
c. 2.0 m cacl2
d. 2.0 m kbr < my choice
8. determine which compound would lower the freezing point of water the most
a. 2.0 m kl
b. 2.0 m k2so4 < my choice
c. 2.0 m nacl
d. 2.0 m kbr
if any are wrong let me know which is the correct answer and why

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:10
Which is true of transition metals when moving from left to right on the periodic table? the d sublevels are not filled across the period. the cation radii become larger across the period. atomic radii increase slightly and then start to decrease. atomic radii decrease slightly and then start to increase. o
Answers: 2

Chemistry, 22.06.2019 02:00
What is the volume occupied by 10.0 dm3 of gas at standard pressure after it has been compressedat constant temputure to 500.0 kpa?
Answers: 1

Chemistry, 22.06.2019 02:50
What is the overall order of reaction for rate = k[no2]2 ? second order 3/2 order third order zero order none of the listed answers are correct
Answers: 3

Chemistry, 22.06.2019 06:00
Compare and contrast physical changes with chemical changes.
Answers: 1
You know the right answer?
Ineed someone to see if my answers are correct!
1. for solutions, colligative properties...
1. for solutions, colligative properties...
Questions

Physics, 09.12.2021 03:50






Mathematics, 09.12.2021 03:50





History, 09.12.2021 03:50

Computers and Technology, 09.12.2021 03:50


Mathematics, 09.12.2021 03:50





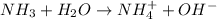
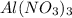 is ionic compounds which dissociated into 4 particles when dissolved in water.
is ionic compounds which dissociated into 4 particles when dissolved in water.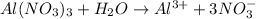
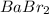 is ionic compounds which dissociated into 3 particles when dissolved in water.
is ionic compounds which dissociated into 3 particles when dissolved in water.