Chemistry, 22.06.2019 19:00 innocentman69
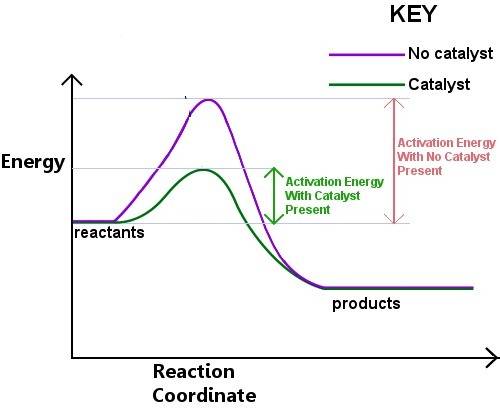
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 05:50
In an exothermic reaction the bonding energy of the product is: less than the reactants same as the reactants greater than the reactants dependent upon the presence of a catalyst
Answers: 1

Chemistry, 22.06.2019 15:00
20 pts ‼️ an unmanned spacecraft travels to mars. mars has a lower strength of gravity than earth. where in the image is the spacecraft’s weight the greatest?
Answers: 1

Chemistry, 23.06.2019 10:00
How many grams of cupric sulfate pentahydrate are needed to prepare 50.00 ml of 0.0800m cuso4× 5h2o?
Answers: 3
You know the right answer?
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated comp...
Questions



Computers and Technology, 13.10.2019 04:50

English, 13.10.2019 04:50


Mathematics, 13.10.2019 04:50

History, 13.10.2019 04:50





Mathematics, 13.10.2019 04:50






Mathematics, 13.10.2019 04:50


English, 13.10.2019 04:50