Chemistry, 23.06.2019 10:00 alejandra216
You dissolve 8.65 grams of lead(l) nitrate in water and then you add 2 50 grams of aluminum. this reaction occurs 2ai(s)+ 3pb(no3)2(aq) -3pb(s)+ 2aino3la(aq) the theoretical yield of solid lead?

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 23.06.2019 00:30
When a beta particle is emitted, the mass number of the nucleus a. decreases by one b. increases by one c. remains the same d. decreases by two
Answers: 2

Chemistry, 23.06.2019 04:40
Equal numbers of moles of he(g), ar(g), and ne(g) are placed in a glass vessel at room temperature. if the vessel has a pinhole-sized leak, which of the following will be true regarding the relative values of the partial pressures of the gases remaining in the vessel after some of the gas mixture has effused?
Answers: 1
You know the right answer?
You dissolve 8.65 grams of lead(l) nitrate in water and then you add 2 50 grams of aluminum. this re...
Questions


Social Studies, 31.07.2019 11:10




History, 31.07.2019 11:10



Mathematics, 31.07.2019 11:10

Chemistry, 31.07.2019 11:10




History, 31.07.2019 11:10

Business, 31.07.2019 11:20

History, 31.07.2019 11:20

Social Studies, 31.07.2019 11:20


Mathematics, 31.07.2019 11:20

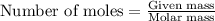
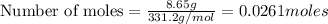
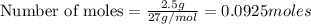
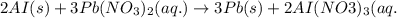
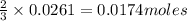 of aluminium.
of aluminium.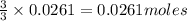 of lead metal.
of lead metal.