
Chemistry, 23.06.2019 13:00 madelyngv97
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole. the δhf for al2o3(s) = -1675.7 kj/mole. finish the equation. δhrxn = [(1)( kj/mole) + (2)( kj/mole)] - [(1)( kj/mole) + (2) ( kj/mole)]

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
Describe the interaction that occurs between two objects with the same electrical charge.
Answers: 1

Chemistry, 22.06.2019 09:20
Give the orbital configuration of the phosphorus (p) atom.
Answers: 1

Chemistry, 22.06.2019 10:00
Water's surface tension and heat storage capacity are accounted for by its a) orbitals b) weight c) hydrogen bonds d) mass e) size
Answers: 2

You know the right answer?
Consider the reaction: 2al(s) + fe2o3(s) → al2o3(s) + 2fe(s) the δhf for fe2o3(s) = -824.3 kj/mole....
Questions

Spanish, 12.11.2020 02:50

English, 12.11.2020 02:50



Social Studies, 12.11.2020 02:50


English, 12.11.2020 02:50


Mathematics, 12.11.2020 02:50


Mathematics, 12.11.2020 02:50

Mathematics, 12.11.2020 02:50

Mathematics, 12.11.2020 02:50

Mathematics, 12.11.2020 02:50


Advanced Placement (AP), 12.11.2020 02:50


Mathematics, 12.11.2020 02:50

Mathematics, 12.11.2020 02:50

Arts, 12.11.2020 02:50

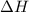 for the given reaction is -851.4 kJ/mol.
for the given reaction is -851.4 kJ/mol.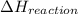
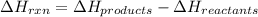
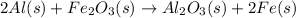
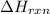 for the reaction is calculated by:
for the reaction is calculated by:![\Delta H_{rxn}=[1(\Delta H_{Al_2O_3})+2(\Delta H_{Fe})]-[1(\Delta H_{Fe_2O_3})+2(\Delta H_{Al})]](/tpl/images/0008/0201/a9d80.png)
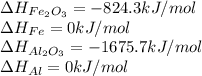
![\Delta H_{rxn}=[1(-1675.7)+2(0)]-[1(-824.3)+2(0)]kJ/mol\\\\\Delta H_{rxn}=-851.4kJ/mol](/tpl/images/0008/0201/8cf6b.png)


