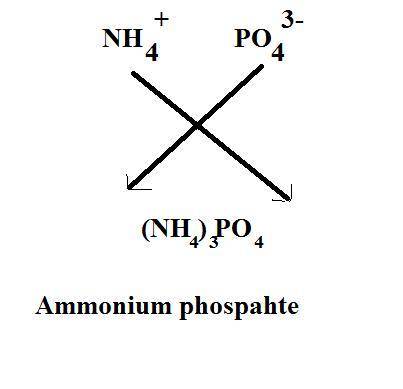
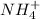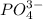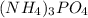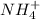What is the chemical formula for the compound ammonium phosphate? use the list of polyatomic ions and the periodic table to you answer. chemical name chemical formula chemical name chemical formula acetate c2h3o2– nitrite no2– carbonate co32– ammonium nh4+ hypocarbonite co2− cyanide cn– hydrogen carbonate (bicarbonate) hco3– hydroxide oh– chlorite clo2– peroxide o22– hypochlorite clo– phosphate po43– chlorate clo3– hydrogen phosphate hpo42– perchlorate clo4– dihydrogen phosphate h2po4– hydronium h3o+ sulfate so42– permanganate mno4– hydrogen sulfate (bisulfate) hso4– nitrate no3– sulfite so32– a. h4h2po4 b. (nh4)2po4 c. (nh4)3po4 d. (nh4)4po4

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:00
For ai it's atomic number is 13 and it's mass number is 27 how many neutrons does it have
Answers: 1

Chemistry, 22.06.2019 01:00
Which type of orbits are found in the principal energy level n = 2 a - s b - s, f c - s, d d - s, p e - s, p, d
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 3

Chemistry, 22.06.2019 09:00
What is the percentage composition of carbon in the compound ch4
Answers: 1
You know the right answer?
What is the chemical formula for the compound ammonium phosphate? use the list of polyatomic ions a...
Questions



Geography, 22.06.2019 10:30

Social Studies, 22.06.2019 10:30





Advanced Placement (AP), 22.06.2019 10:30




Biology, 22.06.2019 10:30

Biology, 22.06.2019 10:30


Mathematics, 22.06.2019 10:30