Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 22.06.2019 07:00
The boiling point of propanoic acid is higher than that of 1-butanol because: propanoic acid has a higher molecular weight than 1-butanol. propanoic acid is more soluble in water than 1-butanol. propanoic acid is a better hydrogen bond donor than 1-butanol. propanoic acid forms hydrogen bonded dimers and 1-butanol does not. 1-butanol forms hydrogen bonded dimers and propanoic acid does not.
Answers: 2


You know the right answer?
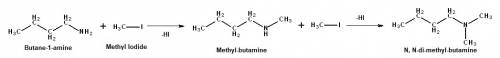
What happens in this reaction? butan-1-amine + ch3i -->...
Questions

Physics, 21.12.2020 19:30

Mathematics, 21.12.2020 19:30





Mathematics, 21.12.2020 19:30

English, 21.12.2020 19:30


World Languages, 21.12.2020 19:30

English, 21.12.2020 19:30

Mathematics, 21.12.2020 19:30


Mathematics, 21.12.2020 19:30



Mathematics, 21.12.2020 19:30

Mathematics, 21.12.2020 19:30

English, 21.12.2020 19:30

Mathematics, 21.12.2020 19:30