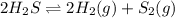Chemistry, 27.06.2019 00:30 cobyontiveros
How does the equilibrium change with the removal of hydrogen (h2) gas from this equation? 2h2s ⇌ 2h2(g) + s2(g) a. equilibrium shifts right to produce more product. b. equilibrium shifts left to produce more reactant. c. equilibrium shifts right to produce less product. d. equilibrium shifts left to produce less reactant.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 05:00
Use the table to identify the phase and phase changes of the elements under the given conditions. write the name of the substance, phase, or phase change
Answers: 3

Chemistry, 22.06.2019 07:40
The formation of a solid, also known as a is an indication of a chemical change. precipitate particulate particle powder
Answers: 3

Chemistry, 22.06.2019 11:40
Modern pennies are composed of zinc coated with copper. a student determines the mass of a penny to be 2.482 g and then makes several scratches in the copper coaling (to expose the underlying zinc). the student puts the scratched penny in hydrochloric acid, where the following reaction occurs between the zinc and the hcl (the copper remains undissolved): zn(s) + 2 hcl(aq) → h2(g) + zncl(aq)the student collects the hydrogen produced over water at 25 °c. the collected gas occupies a volume of 0.899 l at a total pressure of 79 j mmhg. calculate the percent zinc (by mass) in the penny. (assume that all the zn in the penny dissolves.)
Answers: 1

Chemistry, 22.06.2019 16:10
Given the following equation: 2a1 + 3mgcl2 --> 2alcl3 + 3mg how many moles of aluminum chloride are produced from 2.5 moles of magnesium chloride?
Answers: 1
You know the right answer?
How does the equilibrium change with the removal of hydrogen (h2) gas from this equation? 2h2s ⇌ 2h...
Questions


Biology, 05.10.2019 22:00

Physics, 05.10.2019 22:00

Biology, 05.10.2019 22:00





Mathematics, 05.10.2019 22:00

Mathematics, 05.10.2019 22:00




Mathematics, 05.10.2019 22:00

History, 05.10.2019 22:00

English, 05.10.2019 22:00

Arts, 05.10.2019 22:00


Biology, 05.10.2019 22:00