
Chemistry, 28.06.2019 23:00 mercydiaz84
The following chemical equation describes the chemical reaction of hydrogen gas and oxygen gas to create water 2h2 + o2 = 2h2o use what you know about molar relationships to explain how scientists can predict the amount of water produced if they know the amounts of hydrogen and oxygen gases they have to react.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:30
The table describes how some substances were formed substance 19 description formed by boiling pure water formed by combining three hydrogen atoms to every nitrogen atom formed by adding 5 g of sugar to 1 l of water formed by compressing carbon under high pressure based on the given descriptions, which substance is most likely a mixture?
Answers: 1

Chemistry, 22.06.2019 06:00
An alkaline battery produces electrical energy according to the following equation. zn(s) + 2 mno2(s) + h2o(l) zn(oh)2(s) + mn2o3(s) (a) determine the limiting reactant if 17.5 g zn and 31.0 g mno2 are used. (type your answer using the format ch4 for ch4.) (b) determine the mass of zn(oh)2 produced. _ g
Answers: 3


Chemistry, 22.06.2019 14:50
Consider the following multistep reaction: a b→ab(slow) a ab→a2b(fast)−−−−−−−−−−−−−−−−− 2a b→a2b(overall) based on this mechanism, determine the rate law for the overall reaction. express your answer in standard masteringchemistry format. for example, if the rate law is k[a]3[b]2 type k*[a]^3*[b]^2
Answers: 3
You know the right answer?
The following chemical equation describes the chemical reaction of hydrogen gas and oxygen gas to cr...
Questions

Computers and Technology, 02.12.2021 19:30



Business, 02.12.2021 19:30

Mathematics, 02.12.2021 19:30

History, 02.12.2021 19:30


Chemistry, 02.12.2021 19:30


English, 02.12.2021 19:30

Biology, 02.12.2021 19:30

Biology, 02.12.2021 19:30

Advanced Placement (AP), 02.12.2021 19:30

English, 02.12.2021 19:30

Business, 02.12.2021 19:30


Health, 02.12.2021 19:30


English, 02.12.2021 19:30

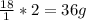 .
.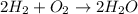
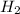 weighs 2 g.
weighs 2 g.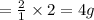
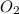 weighs 32 g
weighs 32 g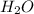 weighs = 18g
weighs = 18g


