
Chemistry, 29.06.2019 05:00 natishtaylor1p8dirz
An atom of sodium-23 (na-23) has a net charge of +1. identify the number of protons, neutrons, and electrons in the atom. then, explain how you determined the number of each type of particle.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 00:10
Think about how you can use le chatelier’s principle to find possible solutions to the design problem. describe at least two ways to increase the yield (amount) of ammonia based on this principle.
Answers: 2

Chemistry, 22.06.2019 10:00
The reactions shown here can be combined to make the overall reaction c(s) + h2o(g) ⇌ co(g) + h2(g) by reversing some and/or dividing all the coefficients by a number. a. c(s) + o2(g) → co2(g) k=1.363×10^69 b. 2 h2(g) + o2(g) → 2 h2o(g) k=1.389×10^80 c. 2co(g) + o2 (g) → 2 co2(g) k=1.477×10^90
Answers: 1

Chemistry, 22.06.2019 10:30
Geothermal energy for industrial use is available almost anywhere. a.true b.false
Answers: 2

You know the right answer?
An atom of sodium-23 (na-23) has a net charge of +1. identify the number of protons, neutrons, and e...
Questions













History, 10.02.2020 22:44

Physics, 10.02.2020 22:44




History, 10.02.2020 22:44

Mathematics, 10.02.2020 22:44

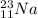
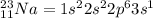
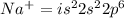 (It will loose one electron)
(It will loose one electron)

