
Chemistry, 29.06.2019 08:00 jameslinimk
For an exothermic reaction at equilibrium, how will increasing the temperature affect keq? a. keq will increase. b. keq will decrease. c. keq will stay the same. d. keq will double.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 11:00
The diagram below shows the different phase transitions that occur in matter. which arrow represents the transition in which dew is formed?
Answers: 1


Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1
You know the right answer?
For an exothermic reaction at equilibrium, how will increasing the temperature affect keq? a. keq w...
Questions

Business, 20.01.2020 08:31





Mathematics, 20.01.2020 08:31

Mathematics, 20.01.2020 08:31

Mathematics, 20.01.2020 08:31


History, 20.01.2020 08:31




History, 20.01.2020 08:31


History, 20.01.2020 08:31




Biology, 20.01.2020 08:31

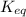 will decrease.
will decrease.![K_{eq} = \frac{[C]^{c}\cdot [D]^d}{[A]^{a} \cdot [B]^{b}}](/tpl/images/0030/3102/48db3.png) , where [A], [B], [C], and [D] are equilibrium concentrations of the four species. Converting some products back to reactants increases [A] and [B] while reducing [C] and [D]. As a result,
, where [A], [B], [C], and [D] are equilibrium concentrations of the four species. Converting some products back to reactants increases [A] and [B] while reducing [C] and [D]. As a result, 

