Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 23:10
Which statement describes both homogeneous mixtures and heterogeneous mixtures?
Answers: 1

Chemistry, 22.06.2019 05:00
1)each group 16 element has how many valence electrons? ( )4 ( )6 ( )8 ( )16 2)how many dots appear in the dot structure for calcium ion, ca2+? ( )zero ( )one ( )two ( )eight 3) which of the following atoms forms a cation to obtain an octet of outer shell electrons? ( )magnesium ( )oxygen ( )fluorine ( )helium 4) an al3+ ion contains 13 protons and 10 electrons. ( )true ( )false 5) valence and non-valence electrons are represented in lewis dot structures. ( )true ( )false
Answers: 3

Chemistry, 22.06.2019 08:00
Me i dont know what to do! the table compares the number of electrons in two unknown neutral atoms. comparison of electrons atom number of electrons a 10 d 11 use this information to determine the number of valence electrons in the atoms. which of the following correctly compares the stability of the two atoms? both are unreactive. both are highly reactive. a is unreactive and d is reactive. a is reactive and d is unreactive.
Answers: 1

Chemistry, 22.06.2019 14:00
8.98 dm3 of hydrogen gas is collected at 38.8 °c. find the volume the gas will occupy at -39.9 °c if the pressure remains constant.
Answers: 3
You know the right answer?
Explain the resonance structures for the nitrate ion, no3−. 1-2 sentences...
Questions



Mathematics, 20.01.2021 01:20



Mathematics, 20.01.2021 01:20


Mathematics, 20.01.2021 01:20

Mathematics, 20.01.2021 01:20

History, 20.01.2021 01:20

Mathematics, 20.01.2021 01:20

Mathematics, 20.01.2021 01:20

Mathematics, 20.01.2021 01:20


Mathematics, 20.01.2021 01:20

Mathematics, 20.01.2021 01:20

Advanced Placement (AP), 20.01.2021 01:20



Computers and Technology, 20.01.2021 01:20

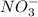 ,have negative charge on overall ion. Negative charge on the one oxygen atom is de-localized to other oxygen atoms present in the molecule of a nitrate ion. Due to this resonance nitrate ions get stabilized.
,have negative charge on overall ion. Negative charge on the one oxygen atom is de-localized to other oxygen atoms present in the molecule of a nitrate ion. Due to this resonance nitrate ions get stabilized.