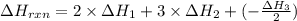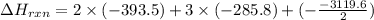Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:40
Determine the energy released per kilogram of fuel used. given mev per reaction, calculate energy in joules per kilogram of reactants. consider 1 mole of tritium plus 1 mole of deuterium to be a mole of “reactions” (total molar mass = 5 grams).
Answers: 1

Chemistry, 22.06.2019 06:00
The tilt of the earth's axis of rotation is responsible for the a) ocean's tides. b) size of the moon. c) brightness of stars. d) earth’s seasons.
Answers: 1

Chemistry, 22.06.2019 10:10
For the reaction, 4 a(g) + 3 b(g) => 2 c(g), the following data were obtained at constant temperature. experiment initial[a],mol/l initial [b],mol/l initial rate,m/min 1 0.200 0.150 5.00 2 0.400 0.150 10.0 3 0.200 0.300 10.0 4 0.400 0.300 20.0 which of the following is the correct rate law for the reaction? 1. rate = k[a]2[b]2 2. rate = k[a][b] 3. rate = k[a]2[b] 4. rate = k[a][b]2
Answers: 3

You know the right answer?
C(graphite) + o2(g) → co2(g)δh o rxn = −393.5 kj/mol h2(g) + 1 2 o2(g) → h2o(l)δh o rxn = −285.8 kj/...
Questions

Mathematics, 21.07.2020 04:01


Mathematics, 21.07.2020 04:01

Health, 21.07.2020 04:01




Mathematics, 21.07.2020 04:01

Health, 21.07.2020 04:01

Mathematics, 21.07.2020 04:01




Mathematics, 21.07.2020 04:01

Mathematics, 21.07.2020 04:01


Mathematics, 21.07.2020 04:01




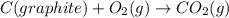
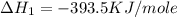
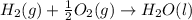
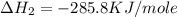
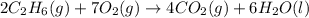
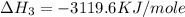
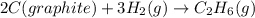
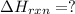
![2\times eq.(1)+\frac{1}{2}(eq.3)\text{[reversing equation 3 and dividing it by 2]}+3(eq.2)](/tpl/images/0034/7108/524c9.png)