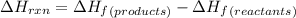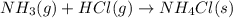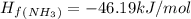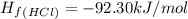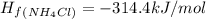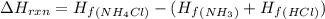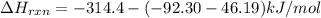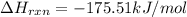Chemistry, 01.07.2019 00:00 genyjoannerubiera
Ammonia (nh3(g), hf = –46.19 kj/mol) reacts with hydrogen chloride (hcl(g), hf = –92.30 kj/mol) to form ammonium chloride (nh4cl(s), hf = –314.4 kj/mol) according to this equation: nh3(g) + hcl(g) nh4cl(s) what is hrxn for this reaction?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 02:30
Which element forms an ionic bond with flourine? 1) fluorine 2) carbon 3) potassium 4) oxygen
Answers: 1

Chemistry, 22.06.2019 06:40
Which statement correctly describes metallic bonds? a. they form when certain atoms lose electrons and other atoms gain electrons. b. they involve an attraction between anions and cations. they always involvpoth a metal and a nonmetal. d. they can only form between atoms of the same element. e. they form because electrons can move freely between atoms.
Answers: 3


Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2
You know the right answer?
Ammonia (nh3(g), hf = –46.19 kj/mol) reacts with hydrogen chloride (hcl(g), hf = –92.30 kj/mol) to f...
Questions


Social Studies, 09.08.2021 20:40



Chemistry, 09.08.2021 20:40

Mathematics, 09.08.2021 20:40

Mathematics, 09.08.2021 20:40

Biology, 09.08.2021 20:40




Mathematics, 09.08.2021 20:40




English, 09.08.2021 20:40




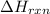 for the given chemical reaction is -175.51 kJ/mol
for the given chemical reaction is -175.51 kJ/mol