Chemistry, 03.07.2019 02:00 710jonathan
Calculate the ph of the solution made by adding 0.50 mol of hobr and 0.30 mol of kobr to 1.00 l of water. the value of ka for hobr is 2.0ă—10â’9. express your answer numerically using two decimal places.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 11:40
Enzymes affect the reactions in living cells by changing the
Answers: 3

Chemistry, 22.06.2019 13:00
In a copper wire, a temperature increase is the result of which of the following
Answers: 1

Chemistry, 23.06.2019 01:00
Which description best characterization the motion of particles in a solid
Answers: 1

Chemistry, 23.06.2019 01:50
Drag the tiles to the correct locations. each tile can be used more than once, but not all tiles will be used. one or more locations will remain empty. nitrosyl fluoride has the chemical formula nof nitrogen has five valence electrons, oxygen has six, and fluorine has seven. complete the lewis structure for this covalent compound. f n = = = . : : 0 : reset next um. all rights reserved us 2
Answers: 2
You know the right answer?
Calculate the ph of the solution made by adding 0.50 mol of hobr and 0.30 mol of kobr to 1.00 l of w...
Questions

Social Studies, 28.11.2020 05:00




Social Studies, 28.11.2020 05:00

Social Studies, 28.11.2020 05:00

History, 28.11.2020 05:10

Biology, 28.11.2020 05:10






Chemistry, 28.11.2020 05:10

Mathematics, 28.11.2020 05:10





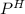 of the solution containing HOBr and KOBr.
of the solution containing HOBr and KOBr.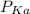 + log
+ log ![\frac{[salt]}{[acid]}](/tpl/images/0044/6662/a0d10.png)
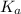 value of HOBr is 2×10⁻⁹.
value of HOBr is 2×10⁻⁹.