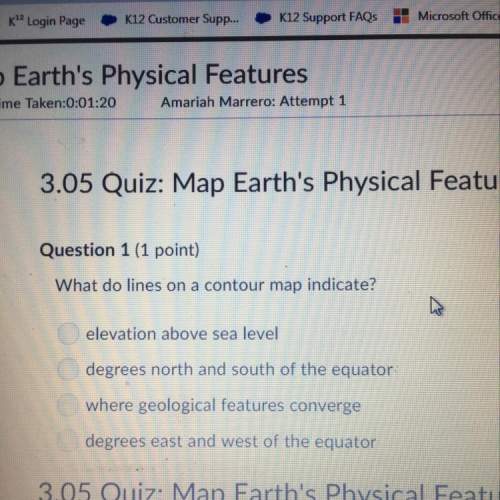
Chemistry, 03.07.2019 23:30 haleyzoey7
How does the law of conservation of mass apply to this reaction: c2h4 + o2 → h2o + co2? a. the equation needs to be balanced. there are fewer oxygen atoms in the equation than hydrogen or carbon. b. only the oxygen needs to be balanced. there are equal numbers of hydrogen and carbon. c. the law of conservation of mass has already been applied. there is an equal number of each element on both sides of the equation. d. each element needs to be balanced.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 07:00
Indicate whether the specified alkyl halides will form primarily substitution products, only elimination products, both substitution and elimination products, or no products when they react with sodium methoxide. 1-bromobutane 1-bromo-2-methylpropane 2-bromobutane 2-bromo-2-methylpropane
Answers: 2


Chemistry, 22.06.2019 15:20
Draw any one of the skeletal structures of a 2° alkyl bromide having the molecular formula of c6h13br and two stereogenic centers. indicate chirality by using wedge and hashed wedge notation. lone pairs do not need to be shown.
Answers: 1

Chemistry, 22.06.2019 18:50
Asample of tin (ii) chloride has a mass of 0.49 g. after heating, it has a mass of 0.41 g. what is the percent by mass of water in the hydrate? %
Answers: 1
You know the right answer?
How does the law of conservation of mass apply to this reaction: c2h4 + o2 → h2o + co2? a. the eq...
Questions


Mathematics, 27.05.2021 04:00



English, 27.05.2021 04:00



Biology, 27.05.2021 04:00



Mathematics, 27.05.2021 04:00


Mathematics, 27.05.2021 04:00






English, 27.05.2021 04:00