
Chemistry, 05.07.2019 06:30 yesman1288
In an experiment, potassium chlorate decomposed according to the following chemical equation. kclo3 → kcl + o2 (molar mass of kclo3 = 122.5 g/mol; kcl = 74.55 g/mol; o2 = 31.998 g/mol) if the mass of kcl produced was 180 grams, which of the following calculations can be used to determine the mass of potassium chlorate decomposed?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:00
What happened in 2012 and how does it illustrate the importance of understanding the sun and how it works?
Answers: 3

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 19:10
Which statement correctly describes the phosphate ion, ? it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms covalently bonded together, and there is a –3 charge on the phosphorus atom. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge distributed over the entire ion. it is composed of one phosphorus atom and four oxygen atoms ionically bonded together, and there is a –3 charge on the phosphorus atom.
Answers: 3

Chemistry, 22.06.2019 22:00
In order to complete this lab. you will need to be familiar with some common chemistry terms. complete the chemical change puzzle and list the relevant terms and their meaning below a.rectant b.product c.supernate
Answers: 3
You know the right answer?
In an experiment, potassium chlorate decomposed according to the following chemical equation. kclo3...
Questions

Mathematics, 14.12.2020 22:20

Mathematics, 14.12.2020 22:20

History, 14.12.2020 22:20

Mathematics, 14.12.2020 22:20








Biology, 14.12.2020 22:20



Social Studies, 14.12.2020 22:20



Chemistry, 14.12.2020 22:20

History, 14.12.2020 22:20

Engineering, 14.12.2020 22:20

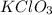 = 122.5 g/mole
= 122.5 g/mole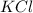 = 74.55 g/mole
= 74.55 g/mole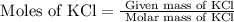
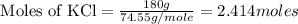
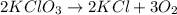
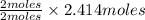 = 2.414 moles of
= 2.414 moles of 

