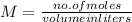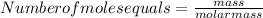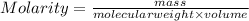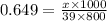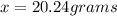Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 16:30
10-14. (a) when 100.0 ml of weak acid ha were titrated with 0.093 81 m naoh, 27.63 ml were required to reach the equivalence point. find the molarity of ha. (b) what is the formal concentration of a- at the equivalence point? (c) the ph at the equivalence point was 10.99. find pk. for ha. (d) what was the ph when only 19.47 ml of naoh had been added?
Answers: 1

Chemistry, 21.06.2019 17:10
There are 6.022 x 10^23 atoms of hg in 1 mole of hg. the number of atoms in 4.5 moles of hg can be found by multiplying 4.5 by 6.022 x 10^23 a. 2.7 x 10^24 b. 27 x 10^23 c. 2.71 x10^24 d. 27.099 x 10^23
Answers: 3

Chemistry, 21.06.2019 22:30
Which of these sequences lists the correct order for the creation of sedimentary rock from sediment? a. deposition, burial, compaction, cementation b. burial, deposition, compaction, cementation c. compaction, deposition, burial, cementation d. cementation, deposition, burial, compaction
Answers: 1

Chemistry, 22.06.2019 12:00
In the following redox reaction which is the oxidizing agent and which is the reducing agent? alcl3 + na nacl + al oxidizing agent = reducing agent =
Answers: 1
You know the right answer?
Acontainer has 800.0 ml of an aqueous solution containing k+ ions. how many grams of k+ ions are in...
Questions

History, 23.09.2019 06:30

Geography, 23.09.2019 06:30


Chemistry, 23.09.2019 06:30

Mathematics, 23.09.2019 06:30

Mathematics, 23.09.2019 06:30

History, 23.09.2019 06:30

English, 23.09.2019 06:30


Physics, 23.09.2019 06:30


English, 23.09.2019 06:30

Mathematics, 23.09.2019 06:30

English, 23.09.2019 06:30

Mathematics, 23.09.2019 06:30

History, 23.09.2019 06:30

Mathematics, 23.09.2019 06:30

Spanish, 23.09.2019 06:30


Mathematics, 23.09.2019 06:30

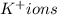 in the solution if the molarity is 0.649 M.
in the solution if the molarity is 0.649 M.