Chemistry, 07.07.2019 18:30 cougar9754
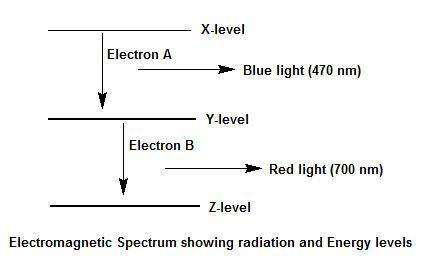
Electron a falls from energy level x to energy level y and releases blue light. electron b falls from energy level y to energy level z and releases red light. which transition, from x to y or from y to z, has a greater energy difference? explain your answer and how you arrived at it. use a diagram of the electromagnetic spectrum.

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 15:20
Select the most likely product for this reaction: koh(aq) + co2(g) – ? k2co3(aq) + h2o(1) k(s) + h2(g) + o2(g) k(s) + co3(9) +h2
Answers: 2


Chemistry, 22.06.2019 19:00
Suppose that a certain fortunate person has a net worth of $71.0 billion ($7.10×1010). if her stock has a good year and gains $3.20 billion (3.20×109) in value, what is her new net worth?
Answers: 3
You know the right answer?
Electron a falls from energy level x to energy level y and releases blue light. electron b falls fro...
Questions


Mathematics, 06.04.2020 23:19

Biology, 06.04.2020 23:19


Mathematics, 06.04.2020 23:19


Mathematics, 06.04.2020 23:19

Biology, 06.04.2020 23:19


Chemistry, 06.04.2020 23:19






Geography, 06.04.2020 23:19

Chemistry, 06.04.2020 23:19


Mathematics, 06.04.2020 23:19

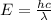
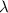 = Wavelength of particle
= Wavelength of particle