Chemistry, 07.07.2019 21:00 christi1175
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with k = 2.80 ✕ ✕ 10−7 sec-1 at 1000°c. cs2(g) → cs(g) + s(g) a. how much of a 4.83-gram sample of carbon disulfide would remain after 37.0 days? 1.97 1.97 grams carbon disulfide b. how much carbon monosulfide would be formed after 37.0 days? 1.14 1.65 grams carbon monosulfide useful information 1.013 bar = 760 torr = 1 atm = 760 mm hg

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 05:00
As you watch a surfer ride a wave towards the shoreline, what is the shoreline? a) displacement reference b) reference point c) coordinate plane d) cartesian boundary
Answers: 1


Chemistry, 22.06.2019 16:00
If 15 drops of ethanol from a medical dropper weight 0.60g, how many drops does it takes from a dropper to dispense 1.0ml of ethanol? the density of ethanol is 0.80g/ml
Answers: 1

Chemistry, 23.06.2019 00:00
How many atoms or molecules are there in a mole of a substance?
Answers: 1
You know the right answer?
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with...
Questions

Mathematics, 24.08.2019 16:00



Mathematics, 24.08.2019 16:00

Chemistry, 24.08.2019 16:00

Mathematics, 24.08.2019 16:00

Chemistry, 24.08.2019 16:00

Mathematics, 24.08.2019 16:00

Chemistry, 24.08.2019 16:00

Mathematics, 24.08.2019 16:00

English, 24.08.2019 16:00

Mathematics, 24.08.2019 16:00

Mathematics, 24.08.2019 16:00


Social Studies, 24.08.2019 16:00



Arts, 24.08.2019 16:00


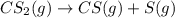
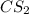 utilised in the reaction
utilised in the reaction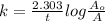
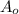 = Initial mass of reactant
= Initial mass of reactant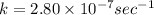
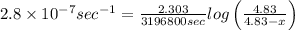
 remained after 37 days = 4.83 - x
remained after 37 days = 4.83 - x