
Chemistry, 08.07.2019 23:00 stefancvorovic1
Asmall diamond, which is made of pure carbon, contains too many carbon atoms to count individually. which is closest to the number of carbon atoms in a 1.0-g diamond? disabled a. 3.6 x 10–45 carbon atoms student selected incorrect b. 9.2 x 101 carbon atoms disabled c. 5.0 x 1022 carbon atoms disabled d. 4,509 carbon atoms

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 17:40
If 10.0 ml of the solution on the right are withdrawn from the 100 ml beaker and diluted again in a similar manner, what is the new concentration? m nacl
Answers: 2

Chemistry, 22.06.2019 17:50
Cryolite, na3alf6(s), an ore used in the production of aluminum, can be synthesized using aluminum oxide. start this question by first balance the chemical equation.1.) balance the equation: - alo3(s)+naoh(l)+hf(> na3alf6+h2o(g). 2.) if 17.5 kilograms of al2o3(s), 51.4 kilograms of naoh(l), and 51.4 kilograms of hf(g) react completely, how many kilograms of cryolite will be produced? 3.)which reactants will be in excess, (al2o3, naoh, or hf) 4.)what is the total mass of the excess reactants left over after the reaction is complete in kg?
Answers: 2

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1

Chemistry, 23.06.2019 00:30
•hydration •dissociation •dissolving which one goes to which
Answers: 1
You know the right answer?
Asmall diamond, which is made of pure carbon, contains too many carbon atoms to count individually....
Questions


Mathematics, 21.08.2019 17:30


Mathematics, 21.08.2019 17:30


English, 21.08.2019 17:30

Mathematics, 21.08.2019 17:30

Social Studies, 21.08.2019 17:30

Social Studies, 21.08.2019 17:30


Mathematics, 21.08.2019 17:30



Computers and Technology, 21.08.2019 17:30



Engineering, 21.08.2019 17:30



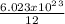 C atoms = 5.02 x 10²² C atoms
C atoms = 5.02 x 10²² C atoms

