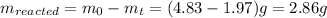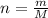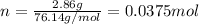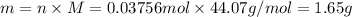Chemistry, 09.07.2019 04:30 romeojose2005
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with k = 2.80 ✕ ✕ 10−7 sec-1 at 1000°c. cs2(g) → cs(g) + s(g) a. how much of a 4.83-gram sample of carbon disulfide would remain after 37.0 days? 1.97 1.97 grams carbon disulfide b. how much carbon monosulfide would be formed after 37.0 days? 1.14 1.65 grams carbon monosulfide useful information 1.013 bar = 760 torr = 1 atm = 760 mm hg

Answers: 1


Another question on Chemistry



Chemistry, 22.06.2019 17:40
Which statement about hf is true? it is zero for any compound in its standard state. it is positive when the bonds of the product store more energy than those of the reactants. it is negative when a compound forms from elements in their standard states. it is zero for any element that is in the liquid state.
Answers: 1

Chemistry, 22.06.2019 22:30
[ou.03jthe pictures below show the wavelengths and intensities of electromagnetic radiations emitted by three stars, star 1, star 2, and star 3. intensity intensity- intensity- 1000 3500 6000 8500 11000 wavelength (a) star 1 1000 3500 6000 8500 11000 1000 3500 6000 8500 11000 wavelength (a) wavelength (a) star 2 star 3 which of these statements is correct about the color of the three stars? star 2 is white in color o star 2 is yellow in color star 1 and star 3 are yellow in color star 1 and star 3 are white in color
Answers: 1
You know the right answer?
The decomposition reaction of carbon disulfide to carbon monosulfide and sulfur is first order with...
Questions




Biology, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01



Mathematics, 20.09.2020 03:01


Medicine, 20.09.2020 03:01




Mathematics, 20.09.2020 03:01


Chemistry, 20.09.2020 03:01

Biology, 20.09.2020 03:01

Mathematics, 20.09.2020 03:01

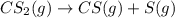
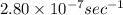 .
.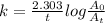
![[A_{0}]](/tpl/images/0068/1392/747e3.png) is initial concentration of reactant and
is initial concentration of reactant and ![[A_{t}]](/tpl/images/0068/1392/b9281.png) is concentration at time t.
is concentration at time t.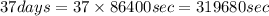
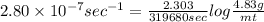
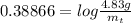
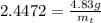
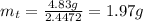
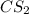 gives 1 mol of CS.
gives 1 mol of CS.