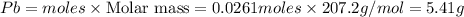You dissolve 8.65 grams of lead(ii) nitrate in water, and then you add 2.50 grams of aluminum. this reaction occurs: 2al(s) + 3pb(no3)2(aq) → 3pb(s) + 2al(no3)3(aq). what’s the theoretical yield of solid lead? use the ideal gas resource and the periodic table. a. 5.41 g b. 11.2 g c. 19.2 g d. 28.8 g

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 03:30
Select the correct answer. when carbon dioxide dissolves in water, it sometimes reacts with water to form carbonic acid as in this balanced equation: co2 + h2o → h2co3. if 495 milliliters of carbon dioxide at 25°c and 101.3 kilopascals reacts with excess water, what is the theoretical yield of carbonic acid? use the periodic table and the ideal gas resource a. 0.889 g b. 1.10g c. 1.27g d. 2.02g what's the answer! quick!
Answers: 1

Chemistry, 22.06.2019 08:30
Which part of earth’s surface receives the most direct rays from the sun? a) equator b) ocean c) poles d) mountains
Answers: 2


Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2
You know the right answer?
You dissolve 8.65 grams of lead(ii) nitrate in water, and then you add 2.50 grams of aluminum. this...
Questions

Social Studies, 02.10.2019 09:30



Mathematics, 02.10.2019 09:30



English, 02.10.2019 09:30






Mathematics, 02.10.2019 09:30

Chemistry, 02.10.2019 09:30

English, 02.10.2019 09:30

Chemistry, 02.10.2019 09:30

Mathematics, 02.10.2019 09:30



Physics, 02.10.2019 09:50

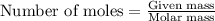
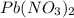

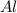
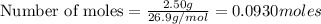
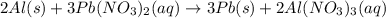
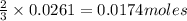 of
of 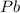
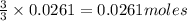 of
of