Chemistry, 10.07.2019 05:00 crisnoobymcnooov72un
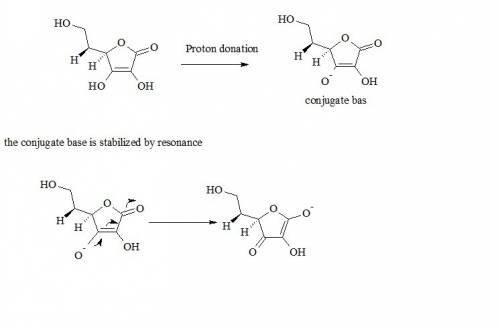
Ascorbic acid (vitamin c. does not contain a traditional carboxylic acid group, but it is, nevertheless, still fairly acidic (pka = 4.2). identify the acidic proton and explain your choice using resonance structures, if necessary:

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
Calculate the h3o+ concentration in a solution of acetic acid if the concentration of molecular acetic acid present at equilibrium is 9.97x10^-3 m and k for the dissociation is 1.86x10^-5. ch3cooh(aq)+h2o(> h3o^+(aq)+ch3coo^-(aq)
Answers: 2

Chemistry, 21.06.2019 22:00
Which describes interactions between substances and stomata during photosynthesis? check all that apply. oxygen enters stomata. oxygen is released through stomata. carbon dioxide enters stomata. carbon dioxide is released through stomata. hydrogen enters stomata. hydrogen is released through stomata.
Answers: 1

Chemistry, 22.06.2019 14:00
How many absorptions would you expect to observe in the 13c nmr spectra of the following molecules? a) 3-chloropentane b) cis-4-methyl-2-pentene
Answers: 2

Chemistry, 22.06.2019 14:30
Which of the following is not one of the steps in the scientific method a. hypothesize b. summarize c. analyze d. familiarize
Answers: 3
You know the right answer?
Ascorbic acid (vitamin c. does not contain a traditional carboxylic acid group, but it is, neverthel...
Questions

Business, 25.04.2020 03:21


History, 25.04.2020 03:21

Mathematics, 25.04.2020 03:21



History, 25.04.2020 03:21


English, 25.04.2020 03:21

Social Studies, 25.04.2020 03:21



English, 25.04.2020 03:21

Business, 25.04.2020 03:21

Mathematics, 25.04.2020 03:21



History, 25.04.2020 03:21