Chemistry, 10.07.2019 13:00 iamsecond235p318rq
What mass of salt (nacl) should you add to 1.42 l of water in an ice cream maker to make a solution that freezes at -11.2 ∘c ? assume complete dissociation of the nacl and density of 1.00 g/ml for water?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:30
Draw the lewis structure for the trisulfur s3 molecule. be sure to include all resonance structures that satisfy the octet rule.
Answers: 3

Chemistry, 22.06.2019 22:20
How do cfcs cause ozone depletion? how do cfcs cause ozone depletion? ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation breaks down cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks two oxygen atoms away from ozone, leaving behind a paired oxygen molecule. ultraviolet radiation creates cfcs, molecules containing chlorine. chlorine then breaks one oxygen atom away from ozone, leaving behind a paired oxygen molecule.
Answers: 2

Chemistry, 23.06.2019 08:00
How many moles of potassium hydroxide are needed to completely react with 2.94 moles of aluminum sulfate
Answers: 1

Chemistry, 23.06.2019 18:30
What is the solution to the problem to the correct number of significant figures (102,900/12)+(170•1.27)
Answers: 1
You know the right answer?
What mass of salt (nacl) should you add to 1.42 l of water in an ice cream maker to make a solution...
Questions


History, 01.08.2019 20:30



Mathematics, 01.08.2019 20:30


History, 01.08.2019 20:30


Business, 01.08.2019 20:30




Business, 01.08.2019 20:30


Business, 01.08.2019 20:30

Mathematics, 01.08.2019 20:30


Mathematics, 01.08.2019 20:30

Mathematics, 01.08.2019 20:30

Spanish, 01.08.2019 20:30

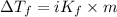
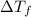 = depression in freezing point
= depression in freezing point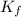 = molal depression constant (
= molal depression constant (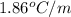 )
)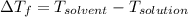
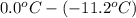
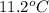
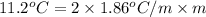 (as sodium chloride dissociate into two ions, i =2)
(as sodium chloride dissociate into two ions, i =2)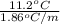
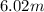
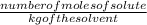
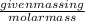
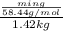 (1 L = 1kg)
(1 L = 1kg)