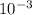Chemistry, 11.07.2019 21:00 keasiabradley
Assume that 1g of aspirin dissolves in 450 ml of water at 10 degrees celsius, how much aspirin would be lost in the 1.5 ml of water added to the reaction mixture if the mixture were at 10 degrees during filtration

Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 10:40
Ammonia and oxygen react to form nitrogen monoxide and water, like this: 4nh3 (g) + 5o2 (g) → 4no (g) + 6h2o (g) also, a chemist finds that at a certain temperature the equilibrium mixture of ammonia, oxygen, nitrogen monoxide, and water has the following composition: compound pressure at equilibrium nh3 65.1atm o2 31.3atm no 62.7atm h2o 65.8atm compound pressure at equilibrium nh3 65.3 atm o2 7.79 atm no 12.1 atm h2o 65.8 atm calculate the value of the equilibrium constant kp for this reaction. round your answer to 2 significant
Answers: 2

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 23:30
Rank substituents in order of their priority when assigning the e or z label to an alkene. i, ch2i , h, ch2ch2cl, f
Answers: 2
You know the right answer?
Assume that 1g of aspirin dissolves in 450 ml of water at 10 degrees celsius, how much aspirin would...
Questions








Business, 30.08.2019 22:20




Health, 30.08.2019 22:20

Mathematics, 30.08.2019 22:20

Mathematics, 30.08.2019 22:20


Mathematics, 30.08.2019 22:20

Geography, 30.08.2019 22:20



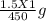 = 3.33 x
= 3.33 x