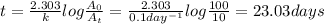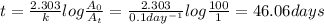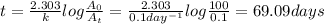Chemistry, 12.07.2019 02:30 devarious83
First-order reaction that results in the destruction of a pollutant has a rate constant of 0.l/day. (a) how many days will it take for 90% of the chemical to be destroyed? (b) how long will it take for 99% of the chemical to be destroyed? (c) how long will it take for 99.9% of the chemical to be destroyed?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 19:50
If a gas has an initial pressure of 101kpa and a volume of 10l, then it expands to a volume of 20l, what is the new pressure?
Answers: 2

Chemistry, 22.06.2019 22:30
Which of the following molecules is polar? c3h7oh c2h5cooh
Answers: 1

Chemistry, 23.06.2019 01:30
Witch two conditions can limit the usefulness of the kinetic molecular theory in describing gas behavior?
Answers: 2

Chemistry, 23.06.2019 05:00
How is electrolysis most commonly used to produce an energy source? a - splitting water molecules produces oxygen, which organisms breathe to fuel their bodies. b - splitting water molecules produces hydrogen gas, which is used to power machines through hydrogen fuel cells. c - splitting carbon dioxide molecules produces coal, a form of carbon that can be burned to produce heat. d - splitting carbon dioxide molecules produces natural gas, which can be burned to generate electricity in power plants.
Answers: 1
You know the right answer?
First-order reaction that results in the destruction of a pollutant has a rate constant of 0.l/day....
Questions



History, 30.08.2019 02:00

Mathematics, 30.08.2019 02:00

Advanced Placement (AP), 30.08.2019 02:00


History, 30.08.2019 02:00


Mathematics, 30.08.2019 02:00


History, 30.08.2019 02:00




History, 30.08.2019 02:00


English, 30.08.2019 02:00


Mathematics, 30.08.2019 02:00

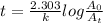
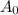 is initial concentration and
is initial concentration and 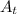 is concentration at time t.
is concentration at time t.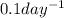 .
.