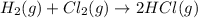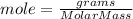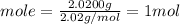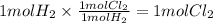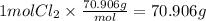Chemistry, 12.07.2019 15:00 inesperezferrere
In an exothermic reaction, chlorine reacts with 2.0200g of hydrogen to form 72.926g of hydrogen chloride gas. how many grams of chlorine reacted with hydrogen

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 20:30
14. complete and balance the equations for the single displacement reactions. a. zn + pb(no3)2 -> b. al + niso4 -> 15. complete and balance the equations for the double displacement reactions. a. agno3(aq) + nacl(aq) -> b. mg(no3)2(aq) + koh(aq) -> 16. complete and balance the equations for the combustion reactions. a. __ ch4 + o2 -> b. __ c3h6 + o2 -> c. + o2 ->
Answers: 2

Chemistry, 22.06.2019 22:40
Percent ionization for a weak acid (ha) is determined by the following formula: percent ionization=[ha] ionized[ha] initial×100%for strong acids, ionization is nearly complete (100%) at most concentrations. however, for weak acids, the percent ionization changes significantly with concentration. the more diluted the acid is, the greater percent ionization.a certain weak acid, ha, has a ka value of 9.4×10? 7.part acalculate the percent ionization of ha in a 0.10 m solution.part bcalculate the percent ionization of ha in a 0.010 m solution
Answers: 1

Chemistry, 23.06.2019 00:00
Which is true about metals used for jewelry, such as platinum and gold? a. they have low flammability. b. they have low reactivity. c. they have high flammability. d. they have high reactivity.
Answers: 1

You know the right answer?
In an exothermic reaction, chlorine reacts with 2.0200g of hydrogen to form 72.926g of hydrogen chlo...
Questions

Mathematics, 14.01.2021 19:00






History, 14.01.2021 19:00




Mathematics, 14.01.2021 19:00

English, 14.01.2021 19:00

Mathematics, 14.01.2021 19:00


Mathematics, 14.01.2021 19:00


Spanish, 14.01.2021 19:00


Mathematics, 14.01.2021 19:00

Mathematics, 14.01.2021 19:00